oq sample size|sample sizes examples : manufacturing The formula used to modify the sample size (calculated the 'standard way' without regard to lot size) is generally taken to be: N/(N+n) where N = lot size and n = sample size.
web2 de nov. de 2022 · Shakhtar Donetsk is going head to head with RB Leipzig starting on 2 Nov 2022 at 17:45 UTC at Stadion Wojska Polskiego w Warszawie stadium, Warsaw city, Poland. The match is a part of the .
{plog:ftitle_list}
webpor benjamin. 18 de junho de 2023. Kamylinha Santos é uma influenciadora digital brasileira conhecida por seus vídeos de dança no TikTok. Nascida em Cajazeiras, .
OQ is not intended to tune-in the parameters. it is intended to validate (or qualify, which is what the Q stands for in OQ) that the parameter settings (input specifications) will produce acceptable parts. The sample sizes may be small or large depending on the type of .
The formula used to modify the sample size (calculated the 'standard way' without .I am trying to determine the sample size needed for Operational Qualification . The answer is- it depends. Depends on what? There are several factors that must be considered when determining the appropriate sample size including risks associated with the product, costs associated with producing .
IQ, OQ, PQ protocols are methods for demonstrating that the equipment being used or installed will offer a high degree of quality assurance such that production processes will consistently manufacture products that . The formula used to modify the sample size (calculated the 'standard way' without regard to lot size) is generally taken to be: N/(N+n) where N = lot size and n = sample size. I am trying to determine the sample size needed for Operational Qualification (OQ). I have ran some parts in the past, thus I can calculate my current mean, std, and CpK etc. .How many people do you need to take your survey? Even if you’re a statistician, determining sample size can be tough. To make it easy, try our sample size calculator. We give you .
and "What are my device's operating limits?". The FDA defines operational qualification as ".establishing confidence that process equipment and sub-systems are capable of . Learn what an Operational Qualification (OQ) is and why you must perform an OQ to manufacture medical devices. We will explain how to establish a process window and running at worst-case settings. You will also learn what .Operational Qualification (OQ): Establishing by objective evidence process control limits and action levels which result in product that meets all predetermined requirements. In another .
If you wanted to see how the opinions of women and men differ (presuming they each make up 50% of the sample), you would wind up with a sample size 139 (1/2 of 278 for each men and women) for a population of 500 (again, half of the total population estimate).Guidance for Industry. 1. Process Validation: General Principles and Practices . This guidance represents the Food and Drug Administration’s (FDA’s) current thinking on this topic. Writing effective IQ/OQ/PQ protocols is a must for following the regulations required by the FDA for equipment, systems, and utilities to demonstrate suitability for the intended use. . Rationale and Sampling .By the end of IQ, OQ and PQ the following should be answered. This will help you understand if your process is stable and capable. Equipment Capability (IQ) Challenge Conditions (OQ) Nominal Operating
The sample size is the maximum expected number of runs (1-alpha% of the time) without a defect for that defect rate. This means that if you have no defects in n runs the defect rate is most likely at or below the stated p value.
Operational Qualification (OQ): Establishing by objective evidence process control limits and action levels which result in product that meets all predetermined requirements. In another word in . Only infinitely large sample size can give us 100% confidence that our measurement coincide with the true value. Naren Patel 34 Reliability .Schoenfeld D. Sample-Size Formula for the Proportional-Hazards Regression-Model. Biometrics. 1983;39(2):499-503. Rubinstein LV, Gail MH, Santner TJ. Planning the duration of a comparative clinical trial with loss to follow-up and a period of continued observation. Introduction to 3Qs of Software IQ-OQ-PQ: IQ, OQ, and PQ constitute the 3Q’s of the Software Validation Process. As testers, we all know that the Software Development Team develops the software in-house as per the Software Requirements Specification (SRS) and Functional Specification, and later the Testing Team verifies the implementation at different .
sample sizes examples
4.2.2 operational qualification (oQ) 4.2.3 Performance qualification (PQ) 4.3 drafting of a validation report 4.4 Formal approval of validation 4.5 Process control and monitoring 4.6 Process changes and revalidation 4.1 Drafting of a validation plan the validation plan should contain, at least, the following details: – iso 11607-1:2009Sample size determination or estimation is the act of choosing the number of observations or replicates to include in a statistical sample.The sample size is an important feature of any empirical study in which the goal is to make inferences about a population from a sample. In practice, the sample size used in a study is usually determined based on the cost, time, or .
Not sure I understand Steven, let me rephrase: We currently use 95% confidence, a reliability requirement based on risk, a previously observed standard dev. and agree upon an acceptable difference of means to get the sample size, through a leading software. Say the output of this is 40. Then say I use the attachment I mentioned to calculate that I need an .
What is IQ, OQ, PQ? These are the abbreviations we use in the medical device industry for the three steps of process validation: Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ).IQ/OQ/PQ ensures that any equipment you use to manufacture your medical device works the way it should—every single time.PQRI Workshop on Sample Sizes for Decision Making in New Manufacturing Paradigms September 13, 2011. 2 Agenda • Process Validation and the FD&C Act • FDA’s Guidance and Statistical Confidence •Satge 1 •Satge 2 •Satge 3. 3 Desired State of ManufacturingInstrument Qualification: A Guide to IQ/OQ Procedures Application note . bandwidth and 0.1 nm step size. This standard is NIST tracable and held in a 1.0 cm quartz cuvette. . If the sample absorbance is above a given limit, or conversely if the
5.4 Operational qualification (OQ) 5.5 Performance qualification (PQ) 6 Maintaining a state of validation 6.1 Monitor and control 6.2 Changes in process and/or product 6.3 Continued state of control 6.4 Examples of reasons for revalidation 7 Use of historical data in process validation 8 Summary of activiti es Annexes In other words, no matter the lot sample size, if one wants to prove with a 95% confidence level that the process is for instance 90% reliable a minimum of 29 sample with no defective (i.e c=number of failure =0). From my .• Size, weight, number of items • e.g. Wrapped complex ortho sets, Major instrument sets in sterilization containers •Assign every set to a family •Within each family, identify the most challenging set •Test the set. 1 Swensen, Donna. Product Quality Assurance Testing: One Hospital’s Experience. Biomedical Engineering and Technology.
The first article in this series, Risk-Based Approaches To Establishing Sample Sizes For Process Validation (June 2016), established the relationship between risk and sample size. Subsequent articles have introduced different methods for determining sample sizes to ensure validation activities will yield valid results. This installment will . As part of process validation through IQ/OQ/PQ, laboratories verify and validate that equipment performs to pre-determined specifications or criteria. ISO/IEC 17025:2017 Validation Requirements ISO/IEC 17025 contains requirements for laboratories, including requirements for equipment used to make measurements within the laboratory. This could be attributed to barrel or screw size, temperature control, water flow, proper mold fill, or any number of equipment or material variables. Documenting the initial installation settings allows for quick identification of the root cause of rejects. Operational Qualification (OQ) The next phase is OQ, or Operational Qualification.Sample Size (n) The size of the sample drawn from the lot. c Acceptance Number. The largest number of nonconforming items from the sample of size n for which the lot should still be accepted. Proportion Defective The proportion of the items of .
This free sample size calculator determines the sample size required to meet a given set of constraints. Also, learn more about population standard deviation.
sample size requirements
o quantitative
Hi guys, I need your expertise for the issue I have with respect to getting the justification on the lot size of the below: Operational Qualification using with the below conditions: Single sampling plan for attribute or pass/fail data with c=0. Sample selected is .
Re-qualifications (OQ or OQ/PQ or OQ/IPV)—provide documented verification that the instrument continues to operate as specified by the manufacturer. Typically performed annually after an initial IQ, OQ, PQ, or IPV has been completed, or according to user’s SOP requirements. . Sample card processing instruments; Instruments other than . Re: Sample size selection for peel strength for variable data using a 95%/99% confide I haven't seen that ISO standard, however, I can confirm that Steven is correct from a couple of sources (which may be cheaper to obtain than buying the ISO standard): Juran's Quality Control Handbook, 4th Edition, discussion on pp. 23.52-56 and table of K values on p.The following components are included on the Procedure Validation Tool for Operational Qualification (OQ) window. Select Power and Sample Size Procedures to Validate Two options are given to allow the user to specify which procedures to validate.,s[ 8s )wxefpmwl 7eqtpi 7m^iw *sv 4vsgiww :epmhexmsr ;lir (iwxvygxmzi 3v )\tirwmzi 8iwxmrk -w 6iuymvih %\ 0dun 'xulydjh )hoorz 7kh iluvw duwlfoh lq wklv vhulhv 5lvn
one quantitative observation
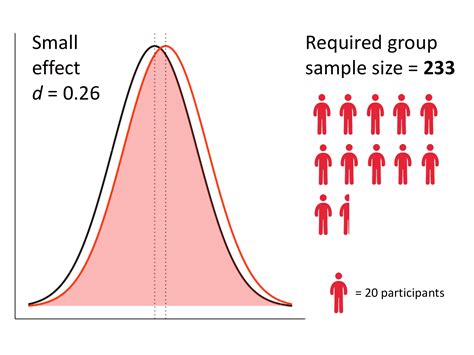
sample size for operational qualification
webObjetivos das Dinâmicas Quebra-Gelo. As dinâmicas quebra-gelo têm vários objetivos e desempenham um papel importante em diferentes contextos. Alguns dos principais .
oq sample size|sample sizes examples